Copper Plating: How Copper Electroplating Works
What are the complex requirements of the process and how can one begin electroplating copper? This post will examine copper plating in-depth and go over some of the common uses for the technique.

What is Copper Electroplating?
Copper electroplating is a straightforward electrochemical method that yields a thin copper coating on any conductive surface through the utilization of an electrolyte bath. The electrolysis process is uncomplicated, with the cathode and anode (positive and negative terminals) attracting opposite charges from both the electrolyte and the anode.
In this process, a pure copper wire serves as the anode, while the product or part requiring electroplating functions as the cathode. The electrolyte solution completes the circuit, facilitating the flow of electricity that allows copper particles to migrate and deposit onto the surface of the cathode.
Despite its apparent simplicity, this setup holds immense advantages for various industries, offering a harmonious blend of qualities. Copper electroplating provides the desirable attributes of copper, including corrosion resistance and conductivity, without compromising the overall strength of the treated product or part.
Key Copper Electroplating Techniques
In essence, copper electroplating necessitates a basic electrolysis setup involving the right combination of anode, cathode, and electrolyte. However, diverse project requirements may demand precise control over the plating rate or the adherence surface, prompting the incorporation of various additives and chemicals. These additives serve as levelers, accelerators, or suppressors, ensuring the attainment of desired results.
Here are the four primary techniques employed by industry experts for copper plating.
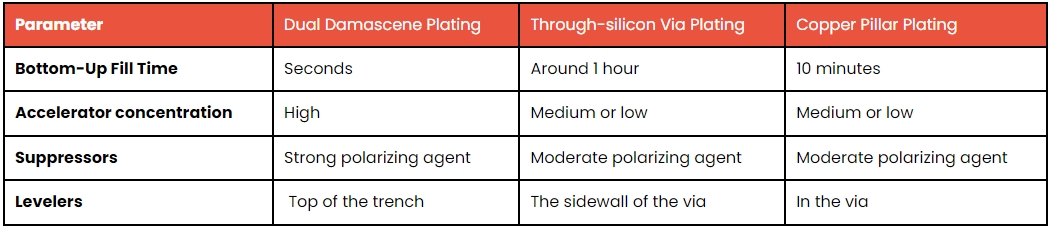
Dual Damascene Plating
Reserved for applications where specific features of the part require prioritized electroplating, the dual damascene technique is commonly utilized for smaller components in the nanometer range and in semiconductors with varying conductivity and resistances for critical applications. In this technique, suppressors, accelerants, and levelers collaborate to achieve bottom-up plating. Suppressants remain on the side walls to diminish the copper deposition rate, while levelers concentrate around the top to reduce the accumulation of copper ions on the surface.
Through-Silicon Via Plating
Similar to the dual damascene method, the through-silicon via plating technique is slower and applied to larger features in the micrometer range. As depicted in the illustration, suppressors and levelers work in tandem to minimize deposition rates on the sides, while accelerators facilitate bottom-up filling. This process is time-consuming, requiring approximately an hour for completion, and the concentration of accelerants on the bottom is kept low.
Copper Pillar Plating
Executing the copper pillar plating process poses a challenge due to its time-consuming and intricate nature. The key objectives here are to ensure the coplanarity of the part and achieve high plating rates without compromising on uniformity.
An effective plating process must yield a precise layer thickness on the surface, necessitating a setup where the part undergoes continuous spinning. This rotational movement, combined with the incoming flow, establishes a consistent current density, ensuring reliable and uniform results.
The following table gives a quick overview of the 3 main aforementioned electroplating techniques.
Redistribution Layer Plating
Exclusive to advanced integrated circuits featuring fan-in and fan-out wafers, the redistribution layer (RDL) plating process boasts precision up to 2 µm, offering exceptional uniformity and reliability.
In RDL applications, a choice exists between the conventional 2D method and the more intricate 3D approach. The 2D method is simpler, as the processing requirements and circuit intricacies are more manageable. Conversely, 3D RDL applications demand precision and optimal process control, contingent upon four critical conditions:
- Wafer Rotation for Uniformity
- Fluid Profile for Optimum Electron Flow
- Tuning and Precision of the Electrical Field
- Use of Additives
Beyond these considerations, the process accommodates additional features in integrated circuits for auxiliary connections and facilitates easier access to new input and output pads. It ensures outstanding uniformity, enhanced purity, and proves to be cost-effective, making it a preferred choice for diverse applications.
Advantages of Copper Electroplating
Discover the five significant benefits of utilizing copper in the electrolysis process:
Flexibility and Durability
Copper’s inherent malleability is a key feature, making it ideal for parts requiring subsequent post-processing. Copper-plated materials can undergo bending, twisting, and various mechanical processes without compromising the surface layer. This not only facilitates operational efficiency but also enhances the lifespan of the treated part.
Excellent Corrosion Protection
Copper exhibits chemical stability and remarkable resistance to corrosion in most conditions. This characteristic renders it an optimal choice for electroplating, as the copper layer provides a protective barrier for the underlying material, particularly if it is susceptible to corrosion. While copper may corrode in severe conditions, it remains a viable option for numerous regular applications.
Great Conductivity
Renowned for its exceptional conductivity in both heat and electricity, copper is a preferred material in the electronics industry. Even if the core metal lacks superior conductivity, the addition of a copper layer imparts this quality, meeting the requirements of various applications.
Excellent Anti-Bacterial Qualities
Copper finds common application in laboratory settings due to its natural ability to resist interaction with bacteria and other microorganisms. This quality eliminates the need for extensive disinfection processes on surfaces or containers with a copper lining.
Cost-Effectiveness
Despite possessing outstanding properties, copper remains among the most economically viable conductive metals globally. Additionally, the electroplating process with a copper sulfate solution is straightforward, further enhancing the cost-effectiveness and practicality of this approach.
Various Types of Copper Electroplating Solutions
The success of electroplating hinges on several factors, including the correct voltage, suitable metal combinations, and an appropriate electrolyte solution containing copper ions. The chemical bonding of these ions significantly influences the capabilities of the electroplating process, offering a choice between alkaline, acidic, or electroless plating baths.
Explore the primary types of copper electroplating solutions utilized in the industry today.
Common Alkaline Copper Baths
Typically, alkaline solutions are favored in applications prioritizing functionality over finesse. Copper electroplating using alkaline baths can be challenging to control and may pose certain health hazards. There are three main types of alkaline baths available for use:
Alkaline Cyanide Solutions
Alkaline solutions containing copper and cyanide are renowned for providing an excellent electroplating coat without functional drawbacks. Generally, copper cyanide solutions ensure a well-adhering and soft coating, facilitating easier buffing and maintenance.
However, the use of cyanide solutions comes with notable drawbacks, including:
Susceptibility to Damage: Most metals used in various applications are prone to damage when exposed to liquids, particularly acids. Alkaline solutions, however, differ in this regard. The cyanide actively protects the base metal until the plating process is complete.
Aesthetic Results: While cyanide solutions yield quick and efficient electroplating results, the aesthetic appeal may be lacking. This process is typically reserved for highly reactive metals, where a thin layer is applied immediately to halt interaction with the environment, followed by additional coatings using a different approach.
Noteworthy drawbacks of using cyanide solutions include:
- Difficult to Control
- Extreme Danger
- Requires Special Handling
- Not Suitable for High Current Density
Despite its drawbacks, the use of alkaline cyanide solutions remains a strategic choice in specific applications where the benefits outweigh the challenges.
Alkaline Solutions Without Cyanide
Cyanide is highly toxic, posing numerous risks for both workers and the environment. Due to its toxicity, many government agencies worldwide prohibit the use of cyanide in any form.
Despite the drawbacks, the advantages of copper solutions based on cyanide are undeniable. Their crucial capability lies in providing essential protection to the base metal for subsequent processing, a feat challenging to replicate with acidic solutions.
In situations where the safety concerns associated with cyanide necessitate an alternative, opting for non-cyanide solutions becomes imperative. Non-cyanide solutions offer a host of advantages, primarily marked by higher current efficiency. These solutions result in a superior appearance, increased stability, and robust leveling power. Additionally, non-cyanide alkaline solutions come at a lower cost, and the overall waste treatment is comparatively more straightforward.
However, it’s worth noting that non-cyanide solutions may not match the effectiveness and efficiency of their cyanide counterparts. Despite this drawback, the evident ease of handling and the less hazardous nature of non-cyanide solutions make them an excellent trade-off in many applications.
Pyrophosphate Copper (Mild Alkalinity)
This solution holds particular significance in applications requiring ductility and exceptional throwing power. Pyrophosphate copper is mildly alkaline and entirely devoid of toxic components, offering a less corrosive alternative. Typically, acidic solutions pose the risk of damaging the base material, while strong alkaline solutions present control challenges. However, this mildly alkaline method strikes a balance, maximizing the benefits of non-corrosiveness without compromising on current density and overall control.
The primary application for electroplating copper using this solution is in the realm of printed circuit boards. It also finds utility in general plating and electroforming. Notably, this process is suitable for materials like plastics, which are typically non-conductive and unsuitable for traditional electroplating methods.
Despite its advantages, it’s crucial to acknowledge that pyrophosphate is considered a pollutant. Therefore, many regional laws mandate proper processing before its disposal, thereby increasing the costs and resources associated with the electroplating process.
Common Acid Copper Baths
Acid solutions present a simpler, more effective, and more precise alternative to alkaline counterparts, offering advantages in cost-effectiveness, versatility, and ease of control. Acid baths can withstand high current densities, resulting in a thicker copper layer—a crucial requirement for applications emphasizing durability.
Two prevalent types of copper surface electroplating solutions are widely used in the industry, employing copper ions in combination with either sulfate or fluoborate ions.
Copper Sulfate Baths
A saturated copper sulfate solution serves as a common electrolyte in the electroplating process. This solution not only facilitates the transfer of electrons but also serves as a copper source for electroplating, eliminating the need for a copper anode. This makes copper sulfate baths ideal for applications requiring direct copper application without frequent anode changes.
The overall cost-effectiveness of copper sulfate baths is notable, as copper sulfate is readily available and cost-effective. The byproducts of the process are harmless, with sulfur deposits at the tank’s bottom, easily extractable and manageable.
However, there are limitations. The sulfate solution must be fully saturated for optimal results, and as the process progresses, copper solution conductivity diminishes due to copper ion loss. To address this, sulfuric acid is commonly added to the solution, maintaining conductivity and acting as a charge carrier.
Copper Fluoborate Baths
Copper fluoborate baths, while less common, operate on similar electrolysis principles. The copper in the solution eliminates the need for anode replacement, and the solution serves as the copper source, akin to copper sulfate.
Notably, copper fluoborate solutions have distinct differences. They exhibit high solubility, allowing a later saturation point, enabling the same volume of solution to hold a greater number of copper ions. This quality is advantageous for high-speed processes or applications requiring coating on large parts, saving time and maintaining process compactness. The higher ion concentration also enhances solution conductivity and provides better current density.
However, there are limitations to copper fluoborate baths. They are comparatively expensive, limiting their widespread use. Additionally, these solutions lack the same throwing power and pose challenges in treatment due to the extreme corrosiveness and hazardous nature of fluoborate ions, contributing to increased overall costs.
Electroless Copper Plating Solution
Electroless baths present a distinct approach. Unlike the aforementioned examples, these baths operate solely on a chemical basis and do not require the passage of electricity for application. This makes them an ideal choice for applications requiring a uniform layer or when dealing with complex geometries. Additionally, electroless baths are capable of handling non-conductive surfaces that would not complete the circuit in a traditional electrolysis setting.
Industrial Applications of Copper Electroplating
Copper electroplating finds widespread applications across diverse industrial sectors owing to its favorable chemical, mechanical, and electrical attributes. Here are the four key industrial areas where copper plays a pivotal role:
Electrical Wiring
A predominant portion of globally processed copper is utilized for electrical wiring, accounting for approximately 60% of copper usage worldwide. Copper’s exceptional conductivity, coupled with its innate malleability, provides electrical wires with the flexibility and transmission capabilities essential for optimal operations.
Electronics
Similar to wiring, electronic components such as circuit boards and relays leverage copper due to its cost-effective suitability for achieving optimal conduction.
Beyond electrical conductivity, copper boasts high heat conduction capabilities. In modern electronics with compact components, copper plating enhances thermal conductivity, serving as a heat sink or aiding heat dissipation to maintain operational temperatures.
Automotive
Copper has long been employed in the automotive sector for wiring, internal components, and various electronic features. The demand for copper in the automotive industry is poised to escalate due to current market trends, particularly the increasing prevalence of electric vehicles.
With electric motors replacing internal combustion engines, copper is gaining relevance in the automobile sector. The solenoids, internal winding, and numerous components within electric motors and their auxiliary systems require a copper layer for durability and optimal performance.
Renewable Energy
Copper, the second most effective metal in conducting electricity after silver (which is prohibitively expensive for large-scale applications), plays a crucial role in renewable energy applications. Copper electroplating is integral for minimizing transmission losses and inefficiencies in renewable energy systems.
Consider solar panels as an illustrative example. Even state-of-the-art solar panels achieve around 23% efficiency, and when factoring in transmission losses due to internal resistance, this figure may decrease. Copper, offering optimal conductivity, emerges as the most ideal choice without significantly impacting overall project costs.
Finishing Services for Metal Parts
Because of its efficiency and adaptability, copper electroplating is the perfect method. Utilizing all of copper’s benefits is possible while maintaining superior mechanical qualities. Furthermore, the copper layer serves as a shield to prevent corrosion and environmental damage to the base metal.
To make sure your parts meet performance and aesthetic requirements, we provide a wide range of metal manufacturing capabilities (CNC machining, metal injection molding, sheet metal fabrication, die casting, and more) and surface finishes.
Our commitment to quality and unwavering focus guarantee accurate and consistent results in the shortest amount of time.
Why do you wait? To begin with our perfect copper electroplating and other manufacturing services, please get in touch with us and provide the technical specifications.
Get a free quote and design analysis today.
We’ll reply to you within 6 working hours.
We respect your privacy.
+86 139 2927 4777 (WhatsApp, Wechat)
